Description
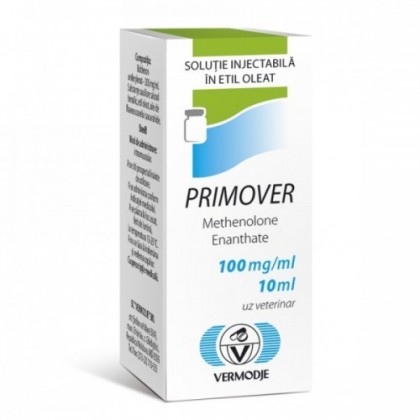
General information about PRIMOVER in USA PRIMOVER (Methenolone Enanthate) is an artificial steroid, similar to the natural testosterone steroid. Each vial contains 10ml and each ml contains 100mg methenelone enanthate. In USA PRIMOVER (Methenolone Enanthate) is a long acting single acting steroid ester. It is the simplest and mildest anabolic steroid. Methenelone enanthate will not aromatize, its non-toxic, and low in androgens. PRIMOVER (Methenolone Enanthate) is used to promote weight gain after extensive surgery, chronic infection, or severe trauma, and in other cases that result in insufficient or maintenance weight gain. PRIMOVER (Methenolone Enanthate) is also used to decrease muscle loss caused by treatment with corticosteroids and to reduce bone pain associated with osteoporosis. PRIMOVER (Methenolone Enanthate) is prescribed as a lean tissue anabolic agent used in cases where muscle loss is evident, such as in post-surgery application. PRIMOVER (Methenolone Enanthate) is generally used as a mild anabolic and anti-catabolic agent as it helps with nitrogen retention. It is a mildly toxic liver with almost no conversion to estrogen. It is generally well tolerated by females. Dosage PRIMOVER (methenolone enanthate) can be taken by both men and women. Dosages for men are 100-300 mg/week, women 1/2 dose. It is the only steroid that works advantageously during a low calorie diet. Effective for bulking, but tends to harden and add muscle tone more than building big muscles. Interactions with other medicinal products in USA PRIMOVER (Methenolone Enanthate) works well when added to a cycle with other steroids, it decreases water retention and hardness when stacked with heavier testosterone injectables, like Omnadren/Sustanon, Cypionate/Propionate, ect .



Reviews
There are no reviews yet.